MIT engineers develop a brand new approach to take away carbon dioxide from air
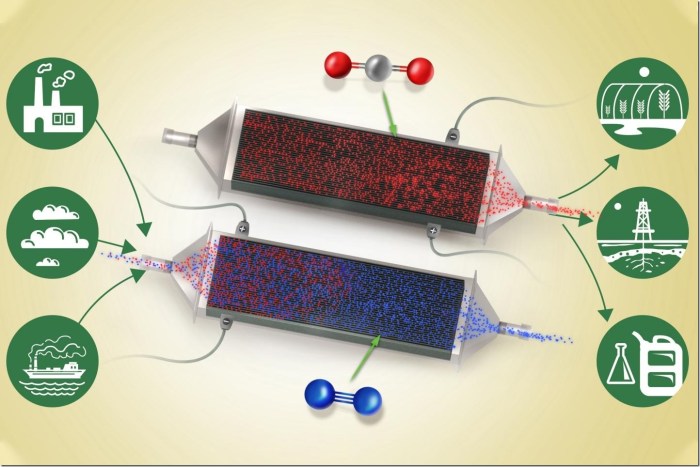
The method might work on the fuel at any concentrations, from energy plant emissions to open air
Massachusetts Institute of Expertise
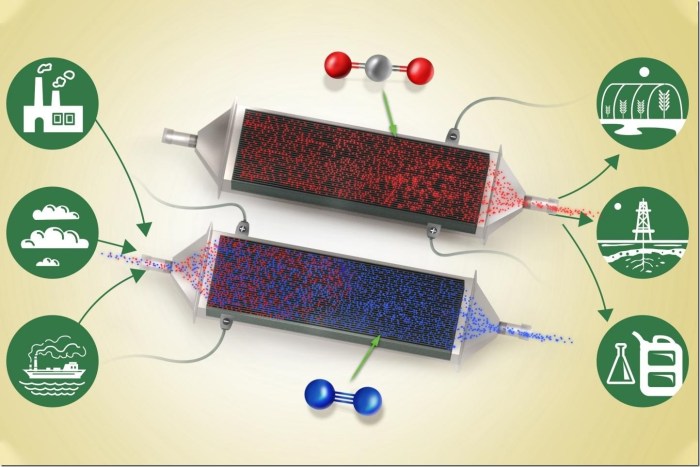
A circulation of air or flue fuel (blue) containing carbon dioxide (crimson) enters the system from the left. Because it passes between the skinny battery electrode plates, carbon dioxide attaches to the charged plates whereas the cleaned airstream passes on by means of and exits at proper. Credit score: Sahag Voskian and T. Alan Hatton
A brand new method of eradicating carbon dioxide from a stream of air might present a big instrument within the battle towards local weather change. The brand new system can work on the fuel at nearly any focus degree, even right down to the roughly 400 elements per million presently discovered within the ambiance.
Most strategies of eradicating carbon dioxide from a stream of fuel require greater concentrations, equivalent to these discovered within the flue emissions from fossil fuel-based energy vegetation. Just a few variations have been developed that may work with the low concentrations present in air, however the brand new technique is considerably much less energy-intensive and costly, the researchers say.
The approach, based mostly on passing air by means of a stack of charged electrochemical plates, is described in a brand new paper within the journal Vitality and Environmental Science, by MIT postdoc Sahag Voskian, who developed the work throughout his PhD, and T. Alan Hatton, the Ralph Landau Professor of Chemical Engineering.
The machine is actually a big, specialised battery that absorbs carbon dioxide from the air (or different fuel stream) passing over its electrodes as it’s being charged up, after which releases the fuel as it’s being discharged. In operation, the machine would merely alternate between charging and discharging, with contemporary air or feed fuel being blown by means of the system throughout the charging cycle, after which the pure, concentrated carbon dioxide being blown out throughout the discharging.
Because the battery expenses, an electrochemical response takes place on the floor of every of a stack of electrodes. These are coated with a compound known as polyanthraquinone, which is composited with carbon nanotubes. The electrodes have a pure affinity for carbon dioxide and readily react with its molecules within the airstream or feed fuel, even when it’s current at very low concentrations. The reverse response takes place when the battery is discharged — throughout which the machine can present a part of the facility wanted for the entire system — and within the course of ejects a stream of pure carbon dioxide. The entire system operates at room temperature and regular air strain.
“The best benefit of this know-how over most different carbon seize or carbon absorbing applied sciences is the binary nature of the adsorbent’s affinity to carbon dioxide,” explains Voskian. In different phrases, the electrode materials, by its nature, “has both a excessive affinity or no affinity in any way,” relying on the battery’s state of charging or discharging. Different reactions used for carbon seize require intermediate chemical processing steps or the enter of serious power equivalent to warmth, or strain variations.
“This binary affinity permits seize of carbon dioxide from any focus, together with 400 elements per million, and permits its launch into any service stream, together with 100 % CO2,” Voskian says. That’s, as any fuel flows by means of the stack of those flat electrochemical cells, throughout the launch step the captured carbon dioxide shall be carried together with it. For instance, if the specified end-product is pure carbon dioxide for use within the carbonation of drinks, then a stream of the pure fuel will be blown by means of the plates. The captured fuel is then launched from the plates and joins the stream.
In some soft-drink bottling vegetation, fossil gasoline is burned to generate the carbon dioxide wanted to present the drinks their fizz. Equally, some farmers burn pure fuel to provide carbon dioxide to feed their vegetation in greenhouses. The brand new system might get rid of that want for fossil fuels in these functions, and within the course of truly be taking the greenhouse fuel proper out of the air, Voskian says. Alternatively, the pure carbon dioxide stream could possibly be compressed and injected underground for long-term disposal, and even made into gasoline by means of a sequence of chemical and electrochemical processes.
The method this method makes use of for capturing and releasing carbon dioxide “is revolutionary” he says. “All of that is at ambient situations — there’s no want for thermal, strain, or chemical enter. It’s simply these very skinny sheets, with each surfaces lively, that may be stacked in a field and related to a supply of electrical energy.”
“In my laboratories, we now have been striving to develop new applied sciences to deal with a spread of environmental points that keep away from the necessity for thermal power sources, modifications in system strain, or addition of chemical substances to finish the separation and launch cycles,” Hatton says. “This carbon dioxide seize know-how is a transparent demonstration of the facility of electrochemical approaches that require solely small swings in voltage to drive the separations.”
In a working plant — for instance, in an influence plant the place exhaust fuel is being produced constantly — two units of such stacks of the electrochemical cells could possibly be arrange facet by facet to function in parallel, with flue fuel being directed first at one set for carbon seize, then diverted to the second set whereas the primary set goes into its discharge cycle. By alternating backwards and forwards, the system might all the time be each capturing and discharging the fuel. Within the lab, the group has confirmed the system can face up to at the least 7,000 charging-discharging cycles, with a 30 % loss in effectivity over that point. The researchers estimate that they’ll readily enhance that to 20,000 to 50,000 cycles.
The electrodes themselves will be manufactured by commonplace chemical processing strategies. Whereas immediately that is performed in a laboratory setting, it may be tailored in order that in the end they could possibly be made in giant portions by means of a roll-to-roll manufacturing course of much like a newspaper printing press, Voskian says. “We now have developed very cost-effective methods,” he says, estimating that it could possibly be produced for one thing like tens of per sq. meter of electrode.
In comparison with different current carbon seize applied sciences, this method is kind of power environment friendly, utilizing about one gigajoule of power per ton of carbon dioxide captured, constantly. Different current strategies have power consumption which differ between 1 to 10 gigajoules per ton, relying on the inlet carbon dioxide focus, Voskian says.
The researchers have arrange an organization known as Verdox to commercialize the method, and hope to develop a pilot-scale plant throughout the subsequent few years, he says. And the system could be very simple to scale up, he says: “If you need extra capability, you simply have to make extra electrodes.”
###
Written by David L. Chandler, MIT Information Workplace
Associated hyperlinks
Paper: “Faradaic Electro-Swing Reactive Adsorption for CO2 Seize.”
https://pubs.rsc.org/en/content material/articlelanding/2019/ee/c9ee02412c#!divAbstract
New kind of electrolyte might improve supercapacitor efficiency
http://information.mit.edu/2019/new-electrolyte-supercapacitor-0812
Eradicating carbon dioxide from energy plant exhaust
http://information.mit.edu/2019/removing-co2-from-power-plant-exhaust-0729
Turning emissions into gasoline
http://information.mit.edu/2017/turning-emissions-into-fuel-1128
MIT researchers develop new approach to clear pollution from water
http://information.mit.edu/2017/electrochemical-clear-pollutants-water-0510
Getting the carbon out of emissions
http://information.mit.edu/2013/getting-the-carbon-out-of-emissions-0625
From EurekAlert!
Like this:
Loading…